New York, Virginia, Maryland each have three illnesses, with one death in Maryland.
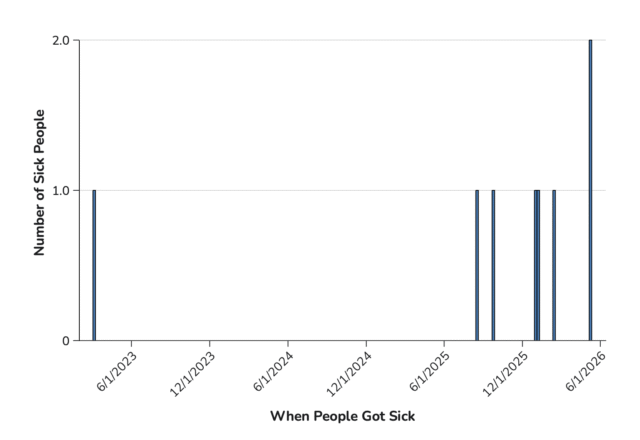
As of June 9, 2026, a total of 9 people infected with the outbreak strain of Listeria have been reported from 3 states. Sick people’s samples were collected on dates ranging from March 6, 2023, to May 10, 2026. Of 9 people with information available, 8 have been hospitalized and 1 death has been reported from Maryland.
Epidemiologic, laboratory, and traceback data show that some requesón cheeses supplied by Clover Hill Dairy are contaminated with Listeria monocytogenes and are making people sick. The investigation is ongoing to determine the source of contamination and whether additional cheeses are linked to the outbreak.
New York State Department of Agriculture and Markets (NYS AGM) collected samples at a retail location shopped at by sick people. One sample of requesón that was repackaged at this retailer tested positive for Listeria monocytogenes. WGS confirmed the Listeria in the repackaged cheese is the same as the Listeria making people sick in this outbreak.
NYS AGM also went to the distributor who supplied cheese to the retail location. They collected a sample from an unopened 18-pound bucket of requesón that was supplied by Clover Hill Dairy. This sample tested positive for Listeria monocytogenes. Whole genome sequencing showed the strain from the cheese is the same strain as the sick people.
Recalled products were distributed in North Carolina, New York, Virginia, Maryland, New Jersey and Washington, DC.
This outbreak may not be limited to the states with known illnesses, and the true number of sick people is likely higher than the number reported. This is because some people recover without medical care and are not tested for Listeria.
Listeria: Marler Clark, The Food Safety Law Firm, is the nation’s leading law firm representing victims of Listeria outbreaks. The Listeria lawyers of Marler Clark have represented thousands of victims of Listeria and other foodborne illness outbreaks and have recovered over $900 million for clients. Marler Clark is the only law firm in the nation with a practice focused exclusively on foodborne illness litigation. Our Listeria lawyers have litigated Listeria cases stemming from outbreaks traced to a variety of foods, such as lettuce, polony, deli meat, cantaloupe, cheese, celery and milk.
If you or a family member became ill with a Listeria infection after consuming food and you’re interested in pursuing a legal claim, contact the Marler Clark Listeria attorneys for a free case evaluation.
Additional Resources: